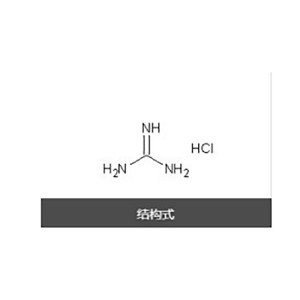
Guanidine hydrochloride
Product name: Guanidine Hydrochloride
Aminoformamidine hydrochloride or Guanidinium chloride
Appearance: white or yellowish lump.
Physical property data
1. Character: white or yellowish lump
2. Melting point (℃): 181-183
3. Relative density (g / ml, 20 / 4 ℃): 1.354
4. Solubility: 228g in 100g water, 76g in 100g methanol and 24g in 100g ethanol at 20 ℃. Almost insoluble in acetone, benzene and ether.
5. PH value (4% aqueous solution, 25 ℃): 6.4
Properties and stability
This product is unstable and can be hydrolyzed into ammonia and urea in aqueous solution, so its toxicity is the same as that of urea. Guanidine and its derivatives are generally more toxic than urea.
Purpose:1. It can be used as intermediate of medicine, pesticide, dye and other organic synthesis. It can be used to synthesize 2-Aminopyrimidine, 2-amino-6-methylpyrimidine and 2-amino-4,6-dimethylpyrimidine. It is an intermediate for the manufacture of sulfadiazine, sulfamethylpyrimidine and sulfadimidine.
2. Guanidine hydrochloride (or guanidine nitrate) reacts with ethyl cyanoacetate to form 2,4-diamino-6-hydroxypyrimidine, which is used to synthesize anti anemia drug folic acid. It can also be used as antistatic agent for synthetic fibers.
3. It can also be used as protein denaturant.
- As a strong denaturant in the experiment of extracting total RNA. Guanidine hydrochloride solution can dissolve protein, cause cell structure damage, nuclear protein secondary structure damage, dissociate from nucleic acid, in addition, RNase can be inactivated by reducing agent such as guanidine hydrochloride.
synthetic method
Using dicyandiamide and ammonium salt (ammonium chloride) as raw materials, the crude guanidine hydrochloride was obtained by melting reaction at 170-230 ℃, and the finished product was obtained by refining.
Contact control
1. Do not inhale dust
2. Harmful if swallowed
3. Eye irritation
4. Skin irritation
Personal protection
1. Wear protective clothing to avoid direct contact or inhalation; 2. Do not drink, eat or smoke at work; 3. Use safety glasses